Product Description UB-221 is a next-generation product developed by Dr. Tse-Wen Chang, the inventor of omalizumab. It is a fully human, original monoclonal antibody formulated for subcutaneous injection. UB-221 not only neutralizes free IgE, which triggers allergic reactions, but also regulates the surface molecule CD23 on B cells, thereby inhibiting IgE production.
Preclinical data have demonstrated that UB-221 possesses superior pharmacological properties compared to Xolair®: UB-221 can neutralize free IgE and also inhibit IgE production by binding to CD23 on B cells (whereas Xolair® only neutralizes free IgE); UB-221 is 8 times more effective than Xolair® in binding affinity to free IgE; in vitro cell experiments show that UB-221 is 2 to 5 times more effective than Xolair® in suppressing IgE production; in monkey studies, UB-221 demonstrated a serum half-life that is twice as long as that of Xolair®.
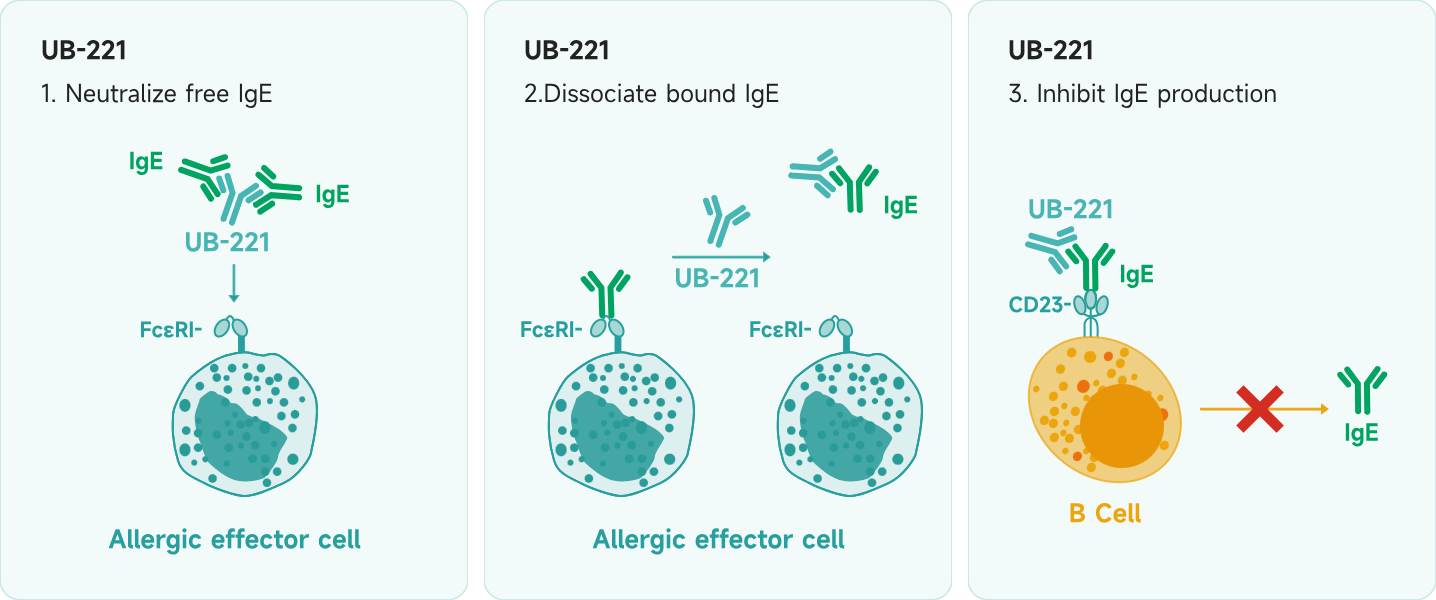






UB-221 is a next-generation product developed by Dr. Tse-Wen Chang, the inventor of omalizumab. It is a fully human, original monoclonal antibody formulated for subcutaneous injection. UB-221 not only neutralizes free IgE, which triggers allergic reactions, but also regulates the surface molecule CD23 on B cells, thereby inhibiting IgE production.
Preclinical data have demonstrated that UB-221 possesses superior pharmacological properties compared to Xolair®:
UB-221 can neutralize free IgE and also inhibit IgE production by binding to CD23 on B cells (whereas Xolair® only neutralizes free IgE);
UB-221 is 8 times more effective than Xolair® in binding affinity to free IgE;
in vitro cell experiments show that UB-221 is 2 to 5 times more effective than Xolair® in suppressing IgE production;
in monkey studies, UB-221 demonstrated a serum half-life that is twice as long as that of Xolair®.
Preclinical data clearly demonstrate that UB-221 has significantly superior pharmacological properties compared to omalizumab:
UB-221 not only neutralizes free IgE but also inhibits IgE production by binding to CD23 on the surface of B cells. Additionally, UB-221 exhibits an 8-fold higher binding affinity to free IgE in humans compared to omalizumab, resulting in more effective neutralization of free IgE.
In vitro cell studies have shown that UB-221 is 2 to 5 times more effective than omalizumab at blocking IgE production. In monkey models, UB-221 displayed a serum half-life twice as long as that of omalizumab, suggesting a longer-lasting therapeutic effect during treatment.
| Limitations of Omalizumab | |
|---|---|
| 1 |
Delayed onset of efficacy leads to slow symptom relief |
| 2 |
Symptoms remain uncontrolled in approximately 50% of patients at therapeutic doses |
| 3 |
Rapid relapse after discontinuation |
| 4 |
High dosing frequency causes inconvenience for patients |
| 5 |
Ineffective in patients with high IgE levels or higher body weight |
| Advantages of UB-221 | |
|---|---|
| 1 |
Visible clinical effect within 1–2 weeks after administration |
| 2 |
Multiple mechanisms of IgE inhibition allow for more effective symptom control |
| 3 |
Greater symptom stability post-treatment, with lower relapse risk |
| 4 |
Reduced dosing frequency improves patients' quality of life |
| 5 |
Greater tolerance to high IgE expression and body weight; efficacy not compromised |