In addition to its potential for achieving functional cure, the monoclonal antibody UB-421 stands out for its remarkable efficacy in treating patients with multidrug-resistant HIV.
The U.S. National Institute of Allergy and Infectious Diseases (NIAID) is currently using UB-421 to treat patients with multidrug-resistant HIV, with highly promising results. These findings were recognized and published in the prestigious journal Nature Medicine in January 2025.
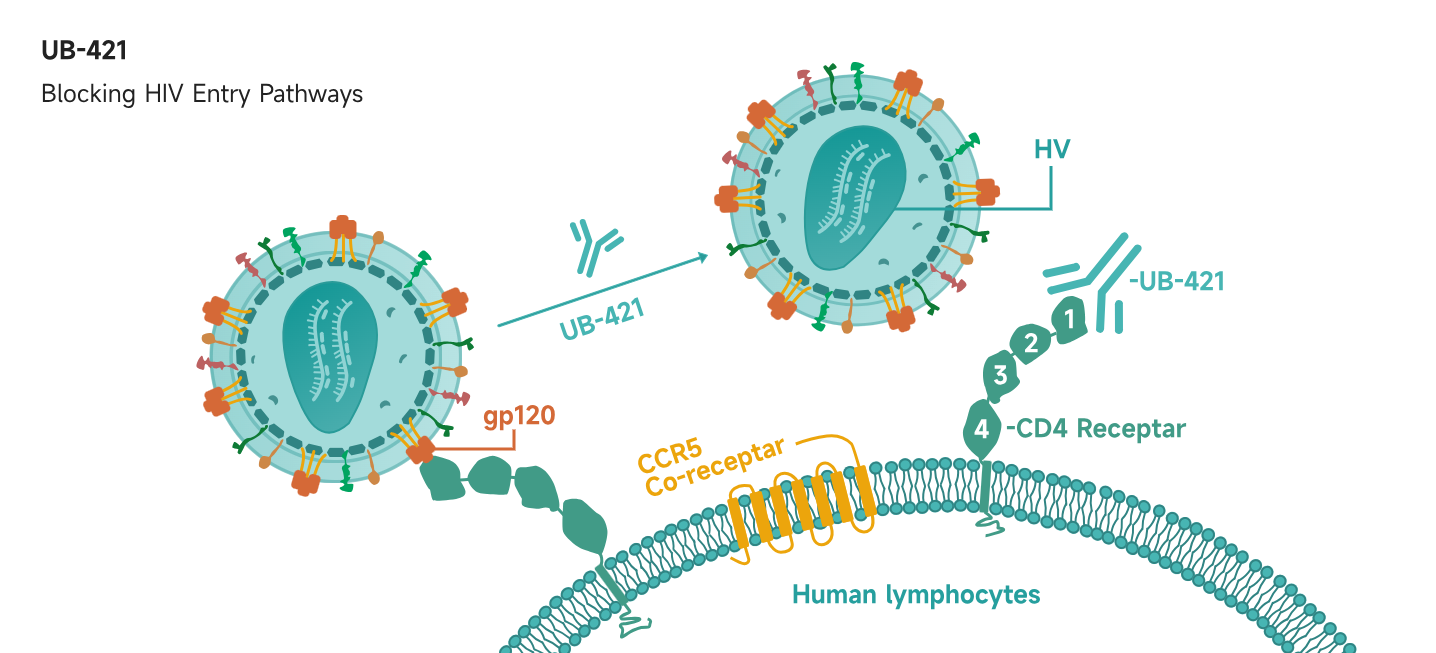







UB-421 not only holds the potential for achieving a functional cure for HIV, but also stands out for its remarkable efficacy in treating patients with multidrug-resistant HIV.
The U.S. National Institute of Allergy and Infectious Diseases (NIAID) is currently using UB-421 to treat patients with multidrug-resistant HIV, with highly significant results. These findings were honored with a publication in the internationally renowned journal Nature Medicine in January 2025.

Single-dose study design
20 treatment-naïve, asymptomatic HIV-positive participants
Two multiple-dose study designs
29 treatment-naïve, asymptomatic HIV-positive participants
Two multiple-dose study designs
Study details completed; focus on evaluating safety, efficacy, and viral suppression in broader patient populations
UB-421 not only holds the potential for achieving a functional cure, but also demonstrates outstanding efficacy in treating patients with multidrug-resistant HIV:
For patients with resistance to existing therapies, UB-421 as a monotherapy has shown significant therapeutic benefits.
Enhances the effectiveness of current treatments by blocking viral replication through multiple mechanisms, enabling effective combination therapy.
Rapidly reduces viral load during the early phase, achieving up to a 99% reduction in blood viral levels. This allows for a staged treatment approach, where UB-421 is combined with other broadly neutralizing antibodies or antiviral agents to strengthen long-term control.