UB-621 is a fully human, original monoclonal antibody formulated for subcutaneous injection, developed for the treatment of human herpes simplex virus (HSV) infections.
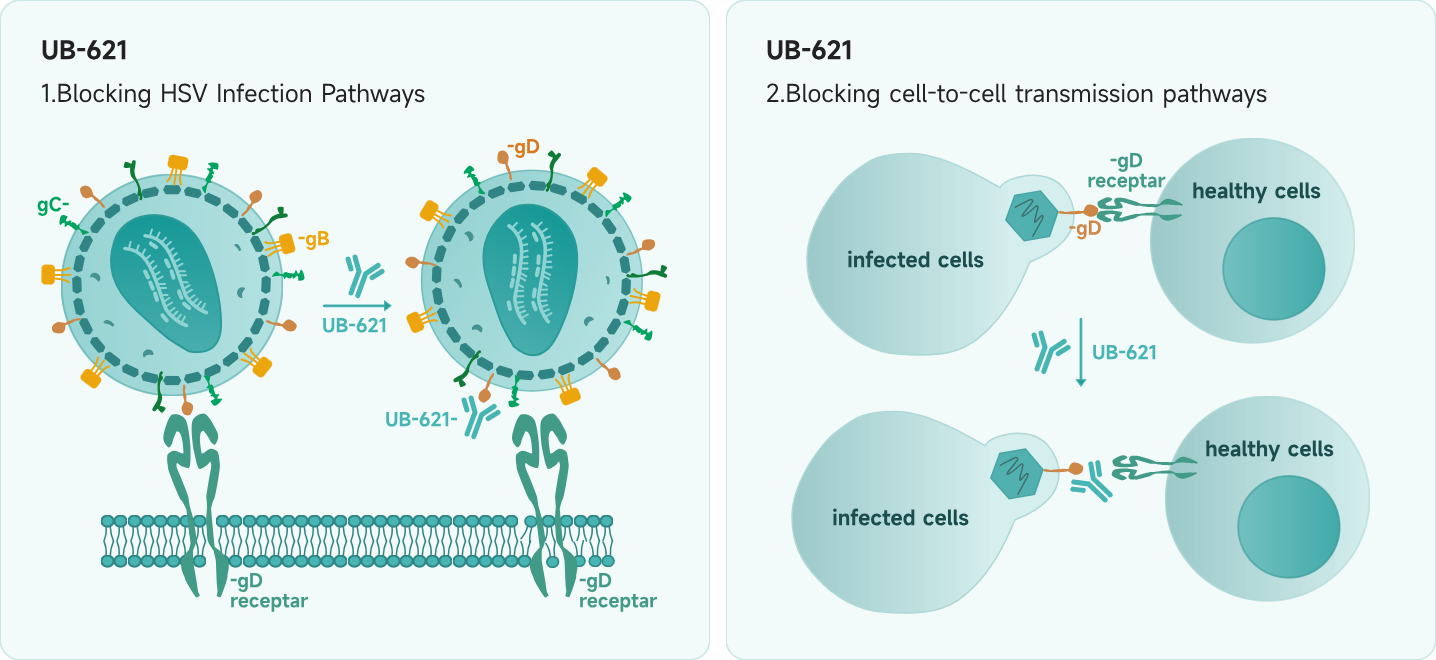
Unlike traditional small-molecule antiviral drugs, UB-621 specifically binds to glycoprotein D on the viral envelope, blocking the interaction between the virus and host cell receptors. This prevents the virus from entering host cells and also disrupts cell-to-cell transmission of HSV. Because UB-621 targets the external pathogen directly, it does not interfere with the body's normal immune function.







UB-621 is a breakthrough innovative monoclonal antibody that makes the suppression of genital herpes recurrence possible.

| Cohort | Clinical Trial Event Summary | |||
|---|---|---|---|---|
| Adverse Events | Serious Adverse Events | Dose-Limiting Toxicity | Anti-Drug Antibodies | |
| Cohort 1 (0.1 mg/kg) |
0 | 0 | 0 | 0 |
| Cohort 2 (0.3 mg/kg) |
0 | 0 | 0 | 0 |
| Cohort 3 (1 mg/kg) |
0 | 0 | 0 | 0 |
| Cohort 4 (3 mg/kg) |
2 | 0 | 0 | 0 |
| Cohort 5 (5 mg/kg) |
1 | 0 | 0 | 0 |
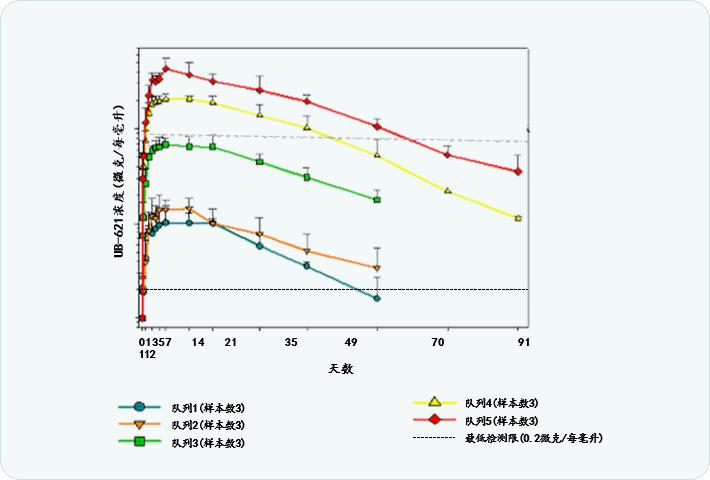
| Dose (mg/kg) |
Half-life (days) |
|---|---|
| 0.1 | 23.2 |
| 0.3 | 28.2 |
| 1 | 26 |
| 3 | 25.7 |
| 5 | 23.5 |